
Thankyou For Subscribing
The latest news and Updates from PurMa Biologics, LLC sent to you inbox
In any serious lab, the 0.22 µm filter is the non-negotiable tool for sterile filtration. It’s what stands between your meticulously prepared cell culture media and the microbial chaos of the outside world. The principle is simple: its pores are engineered to be smaller than the smallest bacteria, physically blocking them from contaminating your precious liquids.
Think of a 0.22 µm filter as a microscopic bouncer at the door of your experiment. It’s an absolute physical barrier that lets the good stuff—your media, buffers, and protein therapeutics—pass through while tossing out the unwanted microbial riff-raff. It's the first and most critical line of defense against the contamination that can derail weeks of work.
That specific 0.22 µm (or 220 nanometers) pore size wasn't just picked out of a hat. It's the result of rigorous scientific validation, pinpointing the exact threshold needed to reliably stop bacteria in their tracks and ensure your work is both safe and reproducible.
For a filter to be officially labeled "sterilizing-grade," it has to pass a tough test. Manufacturers challenge the filter membrane with a massive concentration of a specific, very small bacterium called Brevundimonas diminuta.
Only if the filter successfully removes every last one of these tiny microbes from the liquid does it earn its 0.22 µm rating. This isn't just an academic exercise; it's a critical process for guaranteeing:
The 0.22 µm filter isn't just another piece of lab plastic; it’s an absolute requirement for quality and safety. Its proven ability to produce a sterile filtrate by physically stripping out bacteria is what makes it indispensable everywhere from regulated GMP facilities to discovery labs.
While the main idea is size exclusion—bacteria are simply too big to squeeze through the pores—that's not the whole story. Other forces are at play. Adsorptive effects can also make microbes stick to the filter membrane itself, adding another layer of security to the retention process.
Ultimately, knowing the "why" behind the 0.22 µm standard helps you respect its role in your workflow. You're not just pushing liquid through a membrane; you're running a validated process that underpins your success. Whether you’re prepping media, purifying a protein, or manufacturing a biologic, the 0.22 µm filter is your guarantee of sterility.
Ever wondered what makes a 0.22 µm filter so ridiculously effective at stopping bacteria? It’s not just a simple sieve. A clever combination of physical and chemical forces is at play, ensuring your precious solutions stay sterile. Understanding how this works makes it crystal clear why picking the right filter—and using it correctly—is non-negotiable.
The most obvious mechanism is size exclusion, which is exactly what it sounds like. The filter membrane is a complex, winding maze of pores, each with a nominal diameter of 0.22 micrometers. Most bacteria are simply too big to squeeze through. It's like trying to shove a basketball through the strings of a tennis racket. They get physically blocked.
But what about the bacteria that are smaller than 0.22 µm? Or the ones that can deform under pressure and squish their way through? That's where things get more interesting. Size exclusion isn't the whole story. A second, equally critical mechanism is at work: adsorption.
Filter membranes aren't just inert plastic; they have surface properties that can attract and bind microorganisms. Even if a tiny bacterium is small enough to find a path through a pore, it can get stuck to the internal surfaces of the membrane through weak electrostatic forces. It's captured and prevented from exiting with your filtrate.
This flowchart breaks down the two main pathways for capturing microbes in a filter.
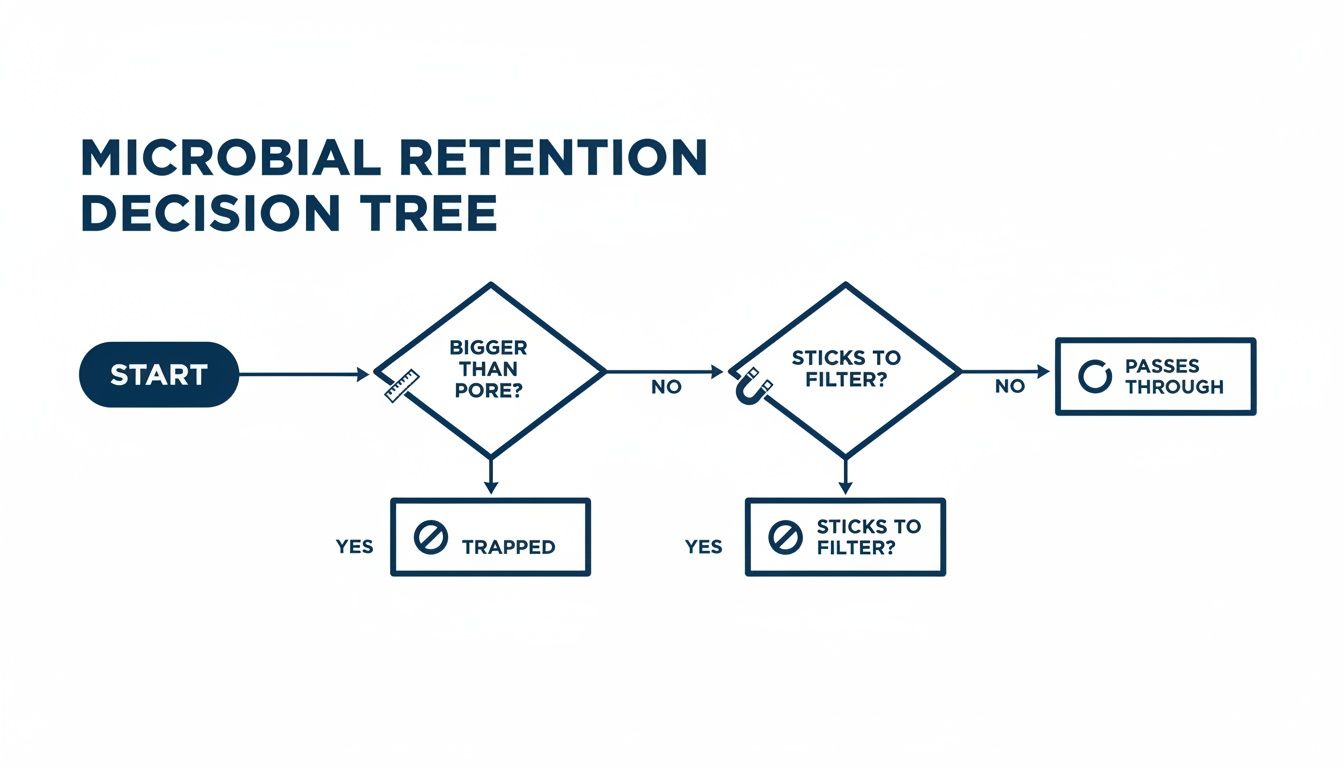
This dual-action defense—physically blocking and chemically grabbing—is what gives a sterilizing-grade filter its high degree of reliability. It’s a layered approach to ensure nothing gets through. To fully appreciate this level of microbial removal, it helps to understand the fundamental difference between disinfecting and sterilizing.
To make this reliability official, regulators and manufacturers validate these filters using a brutal, worst-case scenario. They run a bacterial challenge test using Brevundimonas diminuta (ATCC 19146), one of the smallest known waterborne bacteria. It's the ultimate test of a filter's stopping power.
To be classified as "sterilizing-grade," a filter must achieve a Log Reduction Value (LRV) of at least 7. This means it has to remove a minimum of 99.99999% of the challenge organisms. In practical terms, that’s less than one single bacterium getting through for every ten million you throw at it.
This incredibly tough validation is why the 0.22 µm filter has become the universal standard in biopharmaceutical manufacturing. North America, which accounts for the largest chunk of the sterile filtration market at about 34.07% (a USD 2.71 billion valuation in 2024), depends on this validated technology to maintain quality and satisfy strict regulatory demands.
By combining direct physical blocking with adsorptive capture—and then proving it all against a formidable microbial opponent—the 0.22 µm filter delivers a scientifically-backed guarantee of sterility. This is the solid foundation that underpins the trust that researchers and manufacturers place in it every single day.
Picking the right 0.22 µm filter is a lot like choosing the right tool for a job in the workshop. You wouldn't use a sledgehammer to hang a picture frame. In the lab, matching the filter to the task is a foundational step that directly impacts your workflow’s efficiency, the recovery of your valuable product, and the ultimate success of your experiment.
The sheer variety of options can feel overwhelming, but it all boils down to two simple questions: How much liquid are you filtering, and what’s in it? The answers will point you straight to the perfect filter format and membrane material, ensuring you protect your precious samples without accidentally stripping out what matters most.
This choice is so fundamental that it underpins a massive global industry. The sterile filtration market surpassed USD 7 billion in 2023, and the 0.22 micrometer filtration segment is at the very heart of it. In fact, filters in the 0.1–0.22 µm pore size range are projected to become a USD 6 billion market on their own by 2032. This shows just how central this technology is to modern science. You can explore more about sterile filtration's economic impact for a deeper look at these market dynamics.
The first decision is straightforward: volume. Different filter formats are purpose-built to handle specific liquid quantities. Using the wrong one is a recipe for frustration—wasted time, lost samples, or even a filter blowout in the middle of your prep.
Think of it this way: a syringe filter is for a single espresso shot, a bottle-top filter is for the office coffee pot, and a capsule filter is for the industrial-sized brewer at a café. Each is optimized for its specific scale.
Once you know the format, the next critical choice is the membrane itself. The membrane is the heart of the filter, and its material properties dictate how it interacts with your solution. The big things to consider are protein binding, chemical compatibility, and flow rate.
Choosing the right membrane material is non-negotiable for maximizing recovery and ensuring your filtrate contains what it's supposed to. Here’s a quick rundown of the most common options.
| Membrane Material | Protein Binding | Best For | Chemical Compatibility | Flow Rate |
|---|---|---|---|---|
| PES (Polyethersulfone) | Very Low | Cell culture media, serum, protein solutions | Good with aqueous solutions, weak acids/bases | High |
| PVDF (Polyvinylidene Fluoride) | Low | Protein solutions, biologics, mild organic solvents | Broad; good with alcohols, weak acids | Moderate |
| Nylon | High | Aqueous & organic solutions (NO proteins) | Good with alcohols, bases, most solvents | High |
| PTFE (Polytetrafluoroethylene) | N/A (hydrophobic) | Aggressive solvents, acids, bases, vent filters | Excellent; nearly universal resistance | High |
This table gives you a starting point, but the choice between materials like PES and PVDF for biological work deserves a closer look.
When you’re filtering solutions packed with proteins, growth factors, or antibodies, picking a low-protein-binding membrane is absolutely essential. A material like Nylon, for example, has a high affinity for proteins and will act like a sponge, literally pulling your valuable molecules out of the solution and binding them to the membrane. This leads to significantly lower product recovery and can ruin your experiment before it even starts.
This is where materials specifically engineered for biological applications come in.
By carefully considering both the volume of your liquid and its chemical makeup, you can confidently select a filter 0.22 um that not only guarantees sterility but also maximizes the recovery and integrity of your sample. This thoughtful approach turns filtration from a simple chore into a precise, reliable scientific step.
Let's move from the science of a 0.22 µm filter to the work you do every day at the bench. This small component is the unsung hero of countless workflows, silently protecting your experiments from the costly, time-consuming disaster of contamination. Its role is fundamental to almost every liquid-handling step in cell culture and biologics production.
Whether you're in a small academic lab or a large-scale biopharma facility, the need for absolute sterility is universal. A single bacterium can ruin a culture, compromise a batch, and invalidate weeks of work. Sterile filtration is the non-negotiable step that prevents this, and the 0.22 µm pore size is the industry standard for the job.
One of the most routine yet critical applications is sterilizing basal media and balanced salt solutions. Think of your DMEM, RPMI, or PBS—the foundational liquids that are the very lifeblood of your cells. While many labs purchase pre-sterilized media, you’ll inevitably need to prepare custom formulations or re-sterilize a batch after adding your own supplements.
For this, a bottle-top vacuum filter with a 0.22 µm membrane is the go-to tool. It allows you to efficiently process hundreds of milliliters or even several liters, ensuring every drop that touches your cells is sterile. This isn't just good practice; it's essential for achieving consistent, reproducible cell culture.
This is where filtration really proves its superiority over other sterilization methods like autoclaving. Many of the most important supplements you add to your media are heat-labile, meaning the high temperatures of an autoclave would completely destroy their biological function.
Consider these delicate but essential components:
For these additives, sterile filtration with a small-volume syringe filter is the only viable path. You simply draw up your supplement, attach a 0.22 µm filter, and dispense the sterile, active solution directly into your media. It’s a simple step that preserves the integrity of your most sensitive reagents.
When you're working with high-value biologics, there is zero tolerance for contamination or damage. Filtration is the method of choice for ensuring the sterility of these materials, where preserving the molecule's integrity is just as critical as removing bacteria.
The most significant advantage of a 0.22 µm filter is its ability to achieve sterility at room temperature. For heat-sensitive biologics like fetal bovine serum (FBS), monoclonal antibodies, or viral vectors, autoclaving is not an option—it would irreversibly denature the proteins and render the product useless.
Here are a few critical examples where filtration is indispensable:
In each of these scenarios, the 0.22 µm filter acts as a gentle but absolute gatekeeper. It physically blocks the threat of bacterial contamination without relying on heat or harsh chemicals, preserving the function and value of your most delicate reagents.
Sterile filtration looks simple on the surface, but getting it right every single time is a matter of precise technique. A small slip-up can compromise sterility, tank your product yield, or even blow out the filter membrane entirely. Think of this as your field guide for a smooth, reproducible filtration every time you pick up a syringe.
This isn't just about lab chores; it's a critical quality control step that supports a massive industry. The global sterile filtration market—where the filter 0.22 um is a workhorse—hit USD 8.9 billion in 2023 and is on track to reach USD 15.3 billion by 2028. You can read more about the market growth of sterile filtration to see just how fundamental this process is.
Follow these steps to sidestep the usual mistakes and keep your solution sterile and intact. This method is perfect for small-volume work, like sterilizing media additives, precious protein solutions, or samples for analysis.
Inspect the Packaging First: Before you do anything else, give the sterile blister pack a hard look. If you see any tears, punctures, or signs it’s been compromised, throw the filter away. A breach in the packaging means sterility is already gone.
Attach the Filter Securely: Aseptically pop the 0.22 um filter out of its package. Twist it onto your syringe’s Luer-Lok connection firmly but gently until it’s snug. A loose fit is a classic failure point, letting liquid sneak around the membrane and into your sterile vessel.
Wet the Membrane (Optional but Smart): For aqueous solutions, draw 0.5–1 mL of your solution into the syringe before attaching the filter. Then, attach the filter, point it up, and gently push that small amount of liquid through. This pre-wetting step gets rid of trapped air and ensures you get an even flow from the start.
Apply Steady, Even Pressure: Flip the syringe over and start filtering into your sterile collection container. The absolute key here is gentle and steady pressure. Jamming the plunger down can create a pressure shock that ruptures the delicate membrane, which completely defeats the whole purpose.
Crucial Tip: If the flow rate grinds to a halt, do not just push harder. This is a tell-tale sign the filter is clogged. Forcing it is a surefire way to cause a blowout. Your best move is to stop and switch to a fresh filter.
Even when you do everything right, problems can pop up. Knowing how to diagnose and fix them on the spot will save your sample, your time, and your sanity.
Problem 1: Slow Flow Rate or Clogging
Problem 2: The Dreaded Air Lock
By making this protocol a habit and keeping these troubleshooting tips in your back pocket, you can turn sterile filtration from a potential headache into a predictable, reliable part of your workflow.
In biomanufacturing and clinical research, quality isn't a preference—it's a non-negotiable requirement enforced by regulatory bodies. When you use a 0.22 µm filter, you're not just straining out bacteria; you're executing a critical control point in a much larger quality assurance strategy. But what really makes a filter "validated" and trustworthy for your sterile workflow?
The answer isn't just about the pore size printed on the box. It’s about a rigorous, documented process of testing that happens long before that filter ever arrives at your lab. This assurance starts with the manufacturer, who must prove the filter does exactly what it promises—without introducing any new problems.
A truly validated sterile filter stands on three core pillars of testing: retention, integrity, and safety. Each test answers a different, critical question about the filter’s performance, painting a complete picture of its reliability for use in regulated environments.
Bacterial Challenge Test: This is the ultimate proof. Can the filter actually sterilize a liquid? To find out, the membrane is hit with an incredibly high concentration of a tiny bacterium called Brevundimonas diminuta—at least 10 million organisms per cm² of filter area. To earn the "sterilizing-grade" classification, the 0.22 µm filter has to remove more than 99.99999% of them, a performance metric known as a log reduction value (LRV) of 7.
Integrity Testing: How do you know the filter in your hands isn’t damaged? You can’t see a microscopic tear. That's where integrity tests come in. These are non-destructive checks that confirm the filter's physical structure is intact. The most common is the bubble point test, which finds the exact pressure at which air is first forced through the wetted membrane. This value directly correlates to the filter’s largest pore, confirming there are no defects that could let microbes slip through.
Extractables and Leachables (E&L) Analysis: This is a crucial safety check. Does the filter itself contaminate your sterile fluid? "Extractables" are compounds pulled from the filter material under harsh conditions (like with solvents), while "leachables" are what might migrate into your product under normal use. A validated filter must have an exceptionally low E&L profile to prove it won’t add unwanted chemicals into your media, buffer, or final drug product.
That Certificate of Quality (or Certificate of Analysis) included with your filters is more than just paperwork. It’s your documented proof that the specific lot you're holding has passed these demanding tests.
Think of the Certificate of Quality as your filter's passport. It confirms its identity, proves it has passed all necessary inspections, and grants it permission to be used in your critical process. It is the formal link between the manufacturer's validation work and the filter in your hand.
A trustworthy certificate must provide specific, actionable data, including:
Ultimately, choosing a fully validated 0.22 µm filter from a trusted supplier like PurMa Biologics is fundamental to regulatory compliance and product safety. It provides the documented evidence needed to satisfy auditors and, more importantly, ensures your final product is pure and safe.
Even the most seasoned researchers run into questions at the bench. When it comes to sterile filtration, knowing the “why” behind your tools can be the difference between a clean result and a costly failure.
Here are the straightforward, no-nonsense answers to the most common questions we hear about 0.22 µm filters.
This isn't just about pore size; it's about the job you need to do. Think of a 0.45 µm filter as a bouncer, responsible for clarification or pre-filtration. It does a great job of catching the big stuff—larger debris, precipitates, and most common bacteria—to clean up a solution before the main event.
But a 0.45 µm filter is not sterilizing-grade. Its pores are just big enough to let tiny bacteria, like the notoriously small Brevundimonas diminuta, slip right through. The 0.22 µm filter, however, is the industry-validated standard for sterile filtration. Its pores are small enough to reliably capture these bacteria, giving you a truly sterile final product.
The answer is a hard no. Every sterile 0.22 µm filter is designed, validated, and sold for single use only. Trying to get a second run out of one is a shortcut that will almost certainly ruin your work.
Reusing a sterile filter is a gamble you should never take. The risk of cross-contamination from a previous solution is extremely high, and the filter's structural integrity may be compromised, leading to membrane failure during the second use.
It’s simple: to guarantee sterility and protect the integrity of your process, always discard a filter after one use.
A filter that clogs before your entire solution is through isn't faulty—it's actually doing its job, but it's been completely overwhelmed. This is almost always a sign of a high particulate load in your starting solution. The membrane gets saturated with debris, and liquid can no longer pass through.
Whatever you do, don't just apply more pressure. You'll likely rupture the membrane and compromise sterility. The correct approach is a two-step filtration process:
If you're losing protein after filtration, the culprit is almost always non-specific binding. This is when your valuable protein molecules literally stick to the filter membrane, pulling them out of the solution. This is a particularly notorious problem with certain membrane materials, like nylon.
To stop this from happening, you have to choose a membrane designed for protein work. Always select a low-protein-binding membrane, such as PES (polyethersulfone) or PVDF (polyvinylidene fluoride), when filtering proteins, antibodies, or other biologics. These materials are engineered to ensure your target molecules pass through into the final filtrate, not get stuck behind.
For all your cell culture needs, from validated sterile filters to high-purity media and reagents, trust the experts at PurMa Biologics. Our products are designed to ensure the reliability and reproducibility of your most critical work. Explore our solutions at https://www.purmabiologics.com.

The latest news and Updates from PurMa Biologics, LLC sent to you inbox