Chemically Defined McCoy’s 5A Modified Media (CD-M5AMM)
Chemically Defined McCoy’s 5A Modified Media (CD-M5AMM) does not need any serum , fully synthetic and if free of any trace of animal products. Additionally, the cell growth with CD- McCoy’s 5A is equivalent to Medium+ 10% FBS
Description
Background
PurMa™ Chemically Defined McCoy’s 5A Modified Media (CD-M5AMM)is a proprietary formulated used to grow cells in vitro without any undefined or animal-derived components. Moreover, (CD-M5AMM)contains known quantities of pure chemicals, including amino acids, vitamins, salts, sugars, and more importantly a proprietary combination of synthetic growth factors.
It is absolutely crucial to mention that the PurMa™ Growth Supplement for each conventional media such a DMEM, IMDM, RPMI-1640… (base media ) are slightly different in combination, amount of several growth factors, ions, chemicals, pH with particular adjustments needs to be done, based on the environments the base media provides.
This provides a consistent and controlled environment for cell growth and research. Noticeably, (CD-M5AMM)is essential for applications that require reproducibility and regulatory compliance as well as for studying specific cellular processes. without extraneous variables. Furthermore, (CD-M5AMM)is designed for custom formulations tailored for various cells.
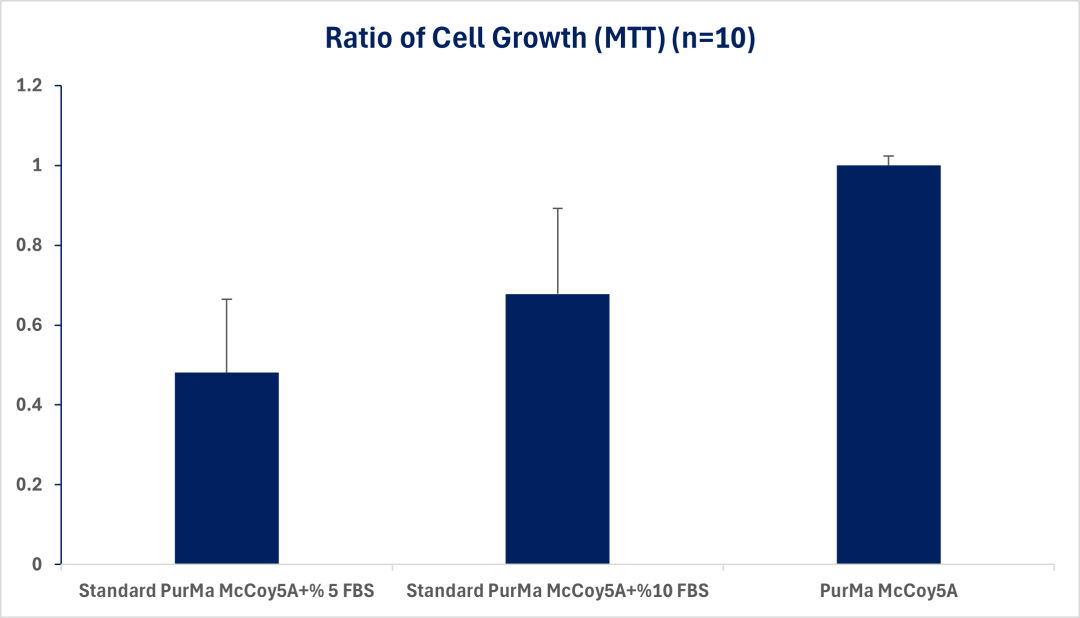
Important Notes:
It is crucially vital for a graduate transition from media containing FBS to chemically defined cell culture media, follow these steps:
- Starting Point: 10% FBS+ (control) (you need two controls, one to be left in only 10% FBS and continue monitoring for comparing with 0% FBS + PurMa™ Chemically Defined Medium. The second one chemically defined medium + 10% FBS which is going through the reduction of FBS process
- 48 hours later: 8% FBS in + PurMa™ Chemically Defined Medium
- 48 hours later: 6% FBS in + PurMa™ Chemically Defined Medium
- 48 hours later: 4% FBS+ PurMa™ Chemically Defined Medium
- 48 hours later: 2% FBS+ PurMa™ Chemically Defined Medium
- 48 hours later: 1 FBS and + PurMa™ Chemically Defined Medium
- 24 hours later: 0$+% FBS and only PurMa™ Chemically Defined Medium
Importantly, for each change, spin down and fully discard the media and replace it with the less percentages of FBS.
Noticeably, you don’t have to start with a huge number of cells (~500,000 cells/ml). Additionally, we strongly recommend you start with more than one plate as some of the plates might get contaminated
So, once the cells are kept in chemically defined Media, you should always keep them in the same chemically defined medium from the same vendor and the same formulation, even for freezing (see below).
Usually, after you get your cells adopted to PurMa™ Chemically Defined Medium you need to make several plates and freeze down several vials. The mentioned cells are adopted to chemically defined medium and when you thaw them in the first week, in addition to PurMa™ Chemically Defined Medium, you need to add 10% PurMa™ CD Booster (Cat# P3C10144).
Formulation
The (CD-M5AMM)includes a base formulation including L-Alanine -L glutamine :Base Formulation of CD McCoy’s 5A P3C310724
Additionally, (CD-M5AMM) contains a proprietary synthetic cocktail of specific growth factors, hormones, and other nutrients which makes it independent of any serum and any trace of animal products.
References
- Altered polyamine metabolism in Chinese hamster cells growing in a defined medium. Sertich GJ, et al. J Cell Physiol. 1986 Apr;127(1):114-20. doi: 10.1002/jcp.1041270115.PMID: 3958058
- Investigations on in vitro survival and virulence of T. pallidum under aerobiosis. Rathlev T. Br J Vener Dis. 1975 Oct;51(5):296-300. doi: 10.1136/sti.51.5.296. PMID: 172190
| Parameter | Specification |
|---|---|
| Appearance | Red, clear liquid |
| pH | 7.2 ± 0.1 |
| Osmolality | 293-355 mOsm/L |
| Endotoxin | NMT< 2EU/mL |
| Mycoplasma | Negative |
| Suitability | Suitable for mammalian cell culture |
| Additive | Sodium pyruvate |
| Indicator | Phenol red |
| Mycoplasma Detection | Negative |
| Sterility Tested | Sterile filtered using 0.0.1 µm filter. 3 Times |
| Form | Liquid |
| Shipping Condition | Room temperature, Over night |
Additional information
| Condition | CD- M5AMM- Standard Formulation, CD- M5AMM Without o L-Alanyl-L-Glutamine, CD- M5AMM -Without Phenol Red, CD- M5AMM -Without Sodium Bicarbonate, CD- M5AMM – Without Glucose, CD- M5AMM – Without Peptone, CD- M5AMM – w/o o L-Alanyl-L-Glutamine, w/o Phenol Red, CD- M5AMM – Without Phenol Red, Without Sodium Bicarbonate, CD- M5AMM – w/ o L-Alanyl-L-Glutamine, w/o Phenol Red, w/o Sodium Bicarbonate, CD- M5AMM – Low Glucose (1.5 g/L), CD- M5AMM – Low Sodium Bicarbonate (0.35 g/L), CD- M5AMM – Low Sodium Bicarbonate (0.85 g/L), CD- M5AMM -15 mM HEPES (3.6 g/L), CD- M5AMM – 25 mM HEPES (5.9 g/L) |
|---|---|
| Size | 1X 100 ml, 1X 1000 ml, 1X 500 ml |
43 reviews for Chemically Defined McCoy’s 5A Modified Media (CD-M5AMM)
Only logged in customers who have purchased this product may leave a review.








azadi@live.ca –
Review: we have purchased Lipid Depleted FBS , DMEM F12 …. they were good quality and decent price
Have you purchased: Yes
Time of submission: 29-Jan-2025 23:10:30
Ease of placing the order: 5/5
Anonymous –
Review:
Have you purchased:
Time of submission: 21-Feb-2025 15:30:00
Ease of placing the order: ‘not given’
Gregory-Mullen@ouhsc.edu –
Review: We buy *all* of our cell culture media, fetal bovine sera, and lipid-depleted sera from PurMa. The quality of their reagents is absolutely top-notch, giving us reliable, repeatable results in our experiments. We highly recommend PurMa!
Have you purchased: Yes
Time of submission: 21-Feb-2025 15:46:22
Ease of placing the order: 5/5
Anonymous –
Review:
Have you purchased:
Time of submission: 24-Feb-2025 13:50:14
Ease of placing the order: ‘not given’
lrothblu@ouhsc.edu –
Review: Serum worked beautifully. Cells grew well and had their usual cycle time. Tissue culture medium also worked very well.
Have you purchased: It is my first Purchase
Time of submission: 24-Feb-2025 13:53:22
Ease of placing the order: 5/5
Anonymous –
Review:
Have you purchased:
Time of submission: 27-Feb-2025 17:42:44
Ease of placing the order: ‘not given’
Anonymous –
Review:
Have you purchased:
Time of submission: 05-Mar-2025 16:31:44
Ease of placing the order: ‘not given’
michael-rudolph@ouhsc.edu –
Review: I have worked directly with PurMa biologics on a number of projects ongoing in my lab. I find the media and the specialty Lipid Depleted Serum to be high quality and very consistent among batches, which is critical to our adipogenesis projects using primary stem cells. Also, the service from PurMa is outstanding for my lab.
Have you purchased: Yes
Time of submission: 05-Mar-2025 16:35:21
Ease of placing the order: 5/5
Anonymous –
Review:
Have you purchased:
Time of submission: 05-Mar-2025 17:29:13
Ease of placing the order: ‘not given’
Anonymous –
Review:
Have you purchased:
Time of submission: 10-Mar-2025 14:32:38
Ease of placing the order: ‘not given’
jkurien@hausbio.com –
Review: We have been working with PurMa biologics for the past several years , we buy cell culture related media like DMEM F12 and EMEM and Fetal Bovine Serum . We appreciate the high quality and reasonable price, and great customer service.
Have you purchased: Yes
Time of submission: 12-Mar-2025 12:34:09
Ease of placing the order: 5/5
hyeungk1968@gmail.comthe –
Review: I used Lipid Free horse serum of PurMa Biologics.
The quality is great!
Have you purchased: Yes
Time of submission: 17-Mar-2025 12:09:58
Ease of placing the order: 5/5
jaya-krishnan@omrf.org –
Review: I started my lab using the regular Gibco media components. We have shifted to Purma past 6 months and see no difference in the quality of our cell culture. Simply saying that Purma is cheaper, quicker to deliver and as good as Gibco. The best part was that Purma customized several media for our fish cell lines which was really helpful. Lastly, Seif walked through the customization of media over Zoom, that gave a lot of confidence for using those media solutions.
Have you purchased: Yes
Time of submission: 17-Mar-2025 14:22:58
Ease of placing the order: 4/5
tayyab-afzal@omrf.org –
Review: We bought Premium FBS and DMEM cell culture media from PurMa Biologics. We are using them for several mammalian cell line and primary cell culture and the cell growth is perfectly fine. We are totally satisfied with the product quality. Importantly as local vendor, we can have the product available as per our demand ASAP.
Have you purchased: Yes
Time of submission: 18-Mar-2025 10:01:50
Ease of placing the order: 5/5
Fitzkr@gmail.com –
Review: We purchase everything we need related to cell and tissue culture from PurMa, reagents like: DMEM, LL15, EMEM, FBS, trypsin, antibiotics mix100X and…
Always great quality, fast processing and shipping.
We are totally satisfied.
Have you purchased: Yes
Time of submission: 22-Mar-2025 10:47:00
Ease of placing the order: 5/5
vmadka@ouhsc.edu –
Review:
Have you purchased: Yes
Time of submission: 24-Mar-2025 15:02:40
Ease of placing the order: 4/5
Anupam-Mandal@ouhsc.edu –
Review: Excellent
Have you purchased: Yes
Time of submission: 24-Mar-2025 15:05:43
Ease of placing the order: 5/5
rohan-varshney@ouhsc.edu –
Review: Reliable and dependable products. We purchased lipid-depleted FBS (LD-FBS) from PurMa Biologics and it worked great. We cross-checked the lipid content in the serum by GC-MS and Triglyceride assay and as advertised, did not find any lipids in the FBS.
Have you purchased: Yes
Time of submission: 24-Mar-2025 15:06:38
Ease of placing the order: 5/5
Surendra-Shukla@ouhsc.edu –
Review: Very fast, efficient, and economic service. Great for new PIs like me.
Have you purchased: Yes
Time of submission: 25-Apr-2025 14:00:04
Ease of placing the order: 5/5
vmadka@ouhsc.edu –
Review:
Have you purchased: Yes
Time of submission: 24-Mar-2025 15:02:40
Ease of placing the order: 4/5
Anupam-Mandal@ouhsc.edu –
Review: Excellent
Have you purchased: Yes
Time of submission: 24-Mar-2025 15:05:43
Ease of placing the order: 5/5
anil-singh@ouhsc.edu –
Review: NA
Have you purchased: Yes
Time of submission: 25-Mar-2025 10:11:14
Ease of placing the order: 5/5
Surendra-Shukla@ouhsc.edu –
Review: Best quality products, best price.
Have you purchased: Yes
Time of submission: 18-Apr-2025 09:51:03
Ease of placing the order: 5/5
madhusmita-rout@ouhsc.edu –
Review:
Have you purchased: Yes
Time of submission: 21-Apr-2025 11:15:46
Ease of placing the order: 5/5
alexholloway@mesogen.com –
Review: This serum appears to be a high quality product and worked well for our needs. Purma has an extensive catalog for cell culturing supplies which we are glad we came across.
Have you purchased: It is my first Purchase
Time of submission: 24-Apr-2025 16:54:29
Ease of placing the order: 5/5
jkurien@hausbio.com –
Review: We have been working with PurMa biologics for the past several years , we buy cell culture related media like DMEM F12 and EMEM and Fetal Bovine Serum . We appreciate the high quality and reasonable price, and great customer service.
Have you purchased: Yes
Time of submission: 12-Mar-2025 12:34:09
Ease of placing the order: 5/5
lrothblu@ouhsc.edu –
Review: Serum worked beautifully. Cells grew well and had their usual cycle time. Tissue culture medium also worked very well.
Have you purchased: It is my first Purchase
Time of submission: 24-Feb-2025 13:53:22
Ease of placing the order: 5/5
hyeungk1968@gmail.comthe –
Review: I used Lipid Free horse serum of PurMa Biologics.
The quality is great!
Have you purchased: Yes
Time of submission: 17-Mar-2025 12:09:58
Ease of placing the order: 5/5
jaya-krishnan@omrf.org –
Review: I started my lab using the regular Gibco media components. We have shifted to Purma past 6 months and see no difference in the quality of our cell culture. Simply saying that Purma is cheaper, quicker to deliver and as good as Gibco. The best part was that Purma customized several media for our fish cell lines which was really helpful. Lastly, Seif walked through the customization of media over Zoom, that gave a lot of confidence for using those media solutions.
Have you purchased: Yes
Time of submission: 17-Mar-2025 14:22:58
Ease of placing the order: 4/5
michael-rudolph@ouhsc.edu –
Review: I have worked directly with PurMa biologics on a number of projects ongoing in my lab. I find the media and the specialty Lipid Depleted Serum to be high quality and very consistent among batches, which is critical to our adipogenesis projects using primary stem cells. Also, the service from PurMa is outstanding for my lab.
Have you purchased: Yes
Time of submission: 05-Mar-2025 16:35:21
Ease of placing the order: 5/5
tayyab-afzal@omrf.org –
Review: We bought Premium FBS and DMEM cell culture media from PurMa Biologics. We are using them for several mammalian cell line and primary cell culture and the cell growth is perfectly fine. We are totally satisfied with the product quality. Importantly as local vendor, we can have the product available as per our demand ASAP.
Have you purchased: Yes
Time of submission: 18-Mar-2025 10:01:50
Ease of placing the order: 5/5
Fitzkr@gmail.com –
Review: We purchase everything we need related to cell and tissue culture from PurMa, reagents like: DMEM, LL15, EMEM, FBS, trypsin, antibiotics mix100X and…
Always great quality, fast processing and shipping.
We are totally satisfied.
Have you purchased: Yes
Time of submission: 22-Mar-2025 10:47:00
Ease of placing the order: 5/5
Gregory-Mullen@ouhsc.edu –
Review: We buy *all* of our cell culture media, fetal bovine sera, and lipid-depleted sera from PurMa. The quality of their reagents is absolutely top-notch, giving us reliable, repeatable results in our experiments. We highly recommend PurMa!
Have you purchased: Yes
Time of submission: 21-Feb-2025 15:46:22
Ease of placing the order: 5/5
azadi@live.ca –
Review: we have purchased Lipid Depleted FBS , DMEM F12 …. they were good quality and decent price
Have you purchased: Yes
Time of submission: 29-Jan-2025 23:10:30
Ease of placing the order: 5/5
Ravi-Gor@ouhsc.edu –
Review: I recently had the pleasure of interacting with Dr. Seif Azadi, and I want to express my gratitude for his expertise and timely assistance. His knowledge and dedication were instrumental in addressing our lab’s needs efficiently.
Have you purchased: It is my first Purchase
Time of submission: 25-Jun-2025 12:00:09
Ease of placing the order: 5/5
xiangpeng-li@ou.edu –
Review: PurMa helped us with the customized medium. They are professional and friendly. I appreciate their great work.
Have you purchased: Yes
Time of submission: 14-Aug-2025 12:59:31
Ease of placing the order: 5/5
jianhua.song@appliedstemcell.com –
Review: it is reliable vendor!
Have you purchased: Yes
Time of submission: 07-Oct-2025 17:14:12
Ease of placing the order: 5/5
Zhenghui.Liu@appliedstemcell.com –
Review: We always buy our agarose from PurMa and have never been disappointed.
Have you purchased: Yes
Time of submission: 08-Oct-2025 11:17:53
Ease of placing the order: 5/5
ravi-gor@ou.edu –
Review: I recently had the pleasure of interacting with Dr. Seif Azadi from Purma Biologics, and I cannot thank him enough for his expertise and prompt assistance. His deep knowledge and dedication were instrumental in meeting our laboratory’s needs with remarkable efficiency.
We ordered FBS, DMEM, and Trypsin from Purma Biologics, and I’m thrilled to report that all the products have performed exceptionally well. They consistently deliver reproducible results, significantly enhancing the reliability of our experiments.
Thank you, Team Purma Biologics, for your good products and support!
Have you purchased: It is my first Purchase
Time of submission: 23-Oct-2025 17:30:25
Ease of placing the order: 5/5
info@takbiotechs.com –
Review: We are extremely satisfied with the quality and consistency of PURMA Biologics products. Both the FBS and cell culture media have delivered excellent performance in our applications, with reliable batch-to-batch consistency. Highly recommended for research and bioprocessing use.
Have you purchased: Yes
Time of submission: 11-Apr-2026 14:49:27
Ease of placing the order: 5/5
aslan.batur@aytakbiotech.com –
Review: We’ve had a great experience with PURMA Biologics products. In particular, their FBS has shown excellent consistency and performance, and their DMEM media has worked very well in our cell culture applications. Overall, very reliable quality.
Have you purchased: Yes
Time of submission: 11-Apr-2026 14:57:39
Ease of placing the order: 5/5
maya.hart@takbiomed.com –
Review: It’s been a pleasure working with PURMA Biologics. Their service is efficient, delivery is fast, and the quality of their products has met our expectations consistently. Very smooth collaboration overall.
Have you purchased: Yes
Time of submission: 11-Apr-2026 15:06:47
Ease of placing the order: 5/5
hajipour@etsu.edu –
Review: PurMa products are of excellent quality and highly reliable.
Have you purchased: Yes
Time of submission: 11-Apr-2026 16:23:59
Ease of placing the order: 5/5